EQUIPE 4 – Wilfried Le Goff et Maryse Guerin – Métabolisme lipidique cellulaire et systémique dans les maladies cardiométaboliques
- Thèmes de recherche
- Membres de l'équipe
- Choix de publications
- Ouvrages
- Collaborations
- Brevets
- Partenaires financiers

Wilfried Le Goff, Team 4 Head
Metabolic diseases are frequently associated with a disturbance of lipid metabolism which may occur at both the systemic and the cell / tissue level. Deregulation of lipid metabolism may involve the liver, which is a key metabolic organ and the major organ in the biogenesis of lipoproteins that transport lipids in the circulation to deliver them in peripheral tissues and cells. Such a disturbance may originate from mutations in key genes controlling circulating lipoprotein levels as observed in familial hypercholesterolemia or from an imbalance in energy sources provided by high-lipid diets enriched in fatty acids and cholesterol – those being strong drivers of metabolic disorders and associated atherosclerotic cardiovascular diseases (ASCVD). After food intake, the first contact of dietary lipids with the organism occurs in the digestive tract, where the microbiota plays a crucial role. Indeed, this first barrier is essential for ensuring well-controlled lipid homeostasis in the whole body. Whatever the source of lipids, when in excess they trigger a large spectrum of deleterious effects which are responsible for the establishment of inflammation, insulin resistance, type 2 diabetes (T2D), metabolic dysfunction associated steatosic liver diseases (MASLD), dyslipidemia and atherosclerosis. The role of these lipids in the development of metabolic disorders is complex and may occur throughout life, from before birth to childhood or adulthood. Molecular and cellular events driving these deleterious effects are not fully understood but involve epigenetic modulation, adaptative gene expression program triggering, production of toxic lipids and rewiring of intracellular lipid metabolism, which impair the metabolic health and function of the targeted tissues.
The main scientific objective of the SLIM research team is to study the alteration of lipid metabolism in rare and frequent diseases associated with a high risk of ASCVD through 3 major axes :
1. To decipher mechanisms causing diet-induced metabolic lipid disorders
2. To explore the link between dynamic lipid remodeling and the risk of ASCVD.
3. To identify new risk factors in ASCVD.
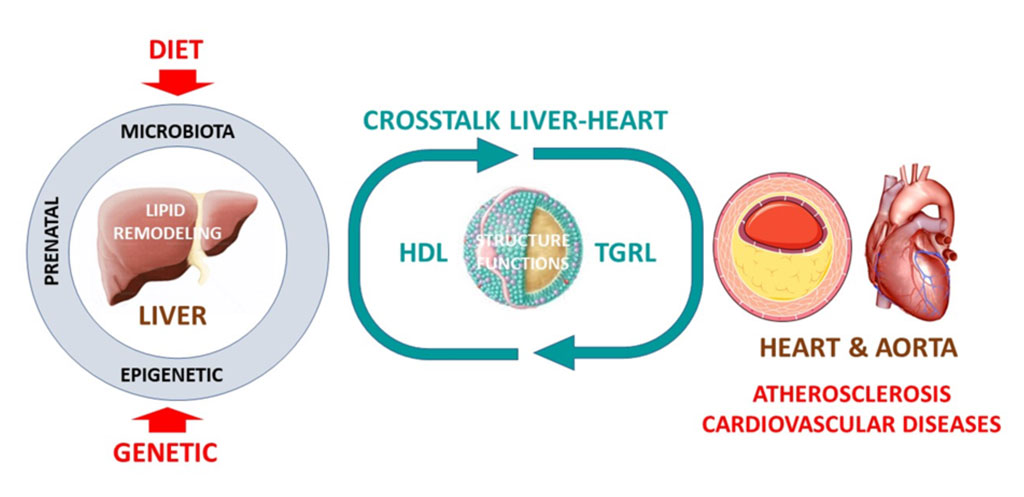
Major research axes. HDL, high-density lipoprotein; TGRL, triglyceride-rich lipoprotein
Recent findings from the SLIM team regarding the 3 research axes.
AXIS 1. To decipher mechanisms causing diet-induced metabolic disorders
A growing body of evidence supports the notion that dietary lipids link diet-induced inflammation and the development of metabolic disorders including MASLD, insulin resistance and T2D. Tissue inflammation is orchestrated by macrophages and their capacity to buffer this excess of lipids plays a major role in tissue inflammation and insulin resistance (PMID: 32752107). Among them, free fatty acids are potent inducers of inflammation and insulin resistance in response to exposure to high-lipid diets. However, while saturated fatty acids trigger macrophage inflammation and promote insulin resistance, polyunsaturated fatty acids, especially omega-3 fatty acids, hamper adipose tissue macrophage (ATM) inflammation and restore insulin sensitivity. In diet-induced obesity, ATM undergoes a metabolic adaptation for which the governing mechanisms are poorly understood. We demonstrated that the membrane ATP-binding cassette G1 (ABCG1) transporter controls the response of ATM to fatty acids carried by triglyceride-rich lipoproteins through the rewiring of saturated fatty acid pools. Importantly, this non-canonical ABCG1-mediated pathway plays a major role in orchestrating ATM plasticity and insulin resistance in diet-induced obesity. (PMID: 39661702).
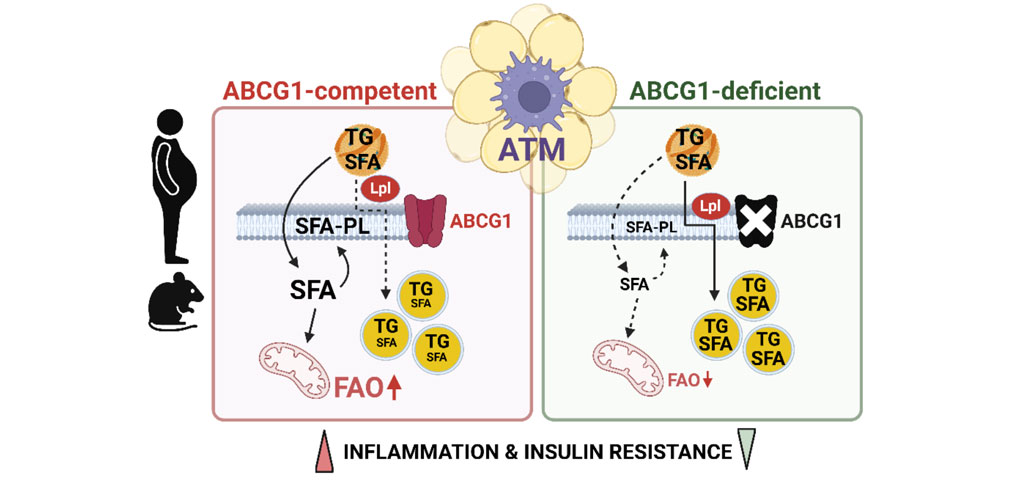
Rewiring of fatty acids by ABCG1 orchestrates the metabolic activation of ATM and contributes to inflammation and insulin resistance in obesity (Dahik V.D. & al., Science Translational Medicine, 2024)
AXIS 2. To explore the link between dynamic lipid remodeling and the risk of ASCVD
HDL structure functions and risk of ASCVD. High-density lipoproteins (HDL) encompass numerous either proteins or lipids that constitute HDL proteome and lipidome, which are intimately link to biological activities of HDL particles. Among more than 300 lipid components of HDL, a sphingolipid namely sphingosine-1-phosphate (S1P) is a bioactive lipid of particular interest in the regulation of cardiovascular diseases. We recently unraveled that the specific composition of plasma and HDL with S1P and their precursors allow to stratify the risk of cardiovascular events in patients with T2D. Our study demonstrates for the first time that patients with T2D exhibited a reduction of circulating S1P species and their precursors which is associated with a high risk to develop cardiovascular diseases. Furthermore, we demonstrated that cardiovascular risk stratification based on CAC score revealed different patterns of S1P species concentration in HDL of T2D patients. Mechanistically, we demonstrated that such an impoverishment of S1P species in HDL leads to impaired anti-inflammatory properties of HDL which might contribute to atherosclerosis development in T2D. Our study provides new important information in the understanding of mechanisms through which alteration of circulating lipidome contributes to CVD in T2D. In addition, it identifies HDL-S1P species and their precursors as key players and potential biomarkers of coronary arterial inflammation in patients with T2D with a high CVD risk (PMID: 39984928)
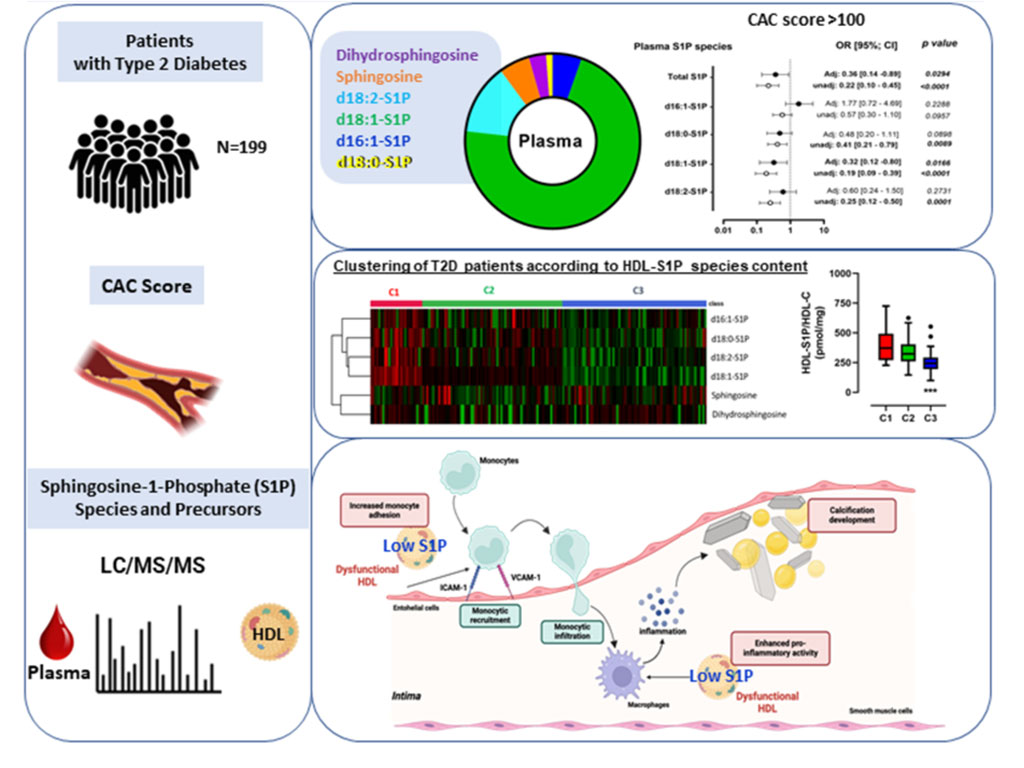
Low HDL-S1P content is associated with a high CAC score and impaired anti-inflammatory activities T2D (Le Goff W. et al., Cardiovascular Diabetology, 2025)
HDL, reverse remnant-cholesterol transport (RRT) and ASCVD. Decreased plasma levels of HDL-C are established as an independent CV risk factor. This association may reflect multiple atheroprotective properties of HDL particles. However, large-scale clinical studies aimed to reduce CV events by raising HDL-C concentrations have failed repeatedly. A large part of HDL-C is derived, in a form of free cholesterol, from triglyceride-rich lipoproteins (TGRL), including chylomicrons and very-low density lipoproteins (VLDL), upon their lipolysis by lipoprotein lipase (LPL). This process is greatly enhanced in the postprandial phase when circulating TG levels are elevated via secretion of chylomicrons but is also active in the interprandial state. Surface fragments (free cholesterol, phospholipids, apolipoproteins) of TGRL liberated during the lipolysis add to the circulating HDL pool; ensuing esterification of free cholesterol by LCAT expands HDL particles with a formation of lipophilic core. Based on these data, we propose a working hypothesis that HDL plays a major role in the both post- and interprandial lipid metabolism, ensuring acquirement and transport of surface fragments generated during lipolysis of TGRL. Such lipid acceptor activity coupled to the transport of TGRL-derived surface lipids (free cholesterol, phospholipid) to the liver may represent a major function of HDL, which is presently overlooked by the mainstream research. As a corollary, HDL-C is an imperfect static measure of this dynamic pathway. In particular, low HDL-C levels associated with elevated CV risk may reflect insufficient cholesterol flux from TGRL to HDL upon lipolysis, resulting in the accumulation of cholesterol in pro-atherogenic TGRL remnants and in the development of CV disease. Lipid acceptor activity of HDL should therefore be diminished in low HDL-C subjects at elevated CV risk.
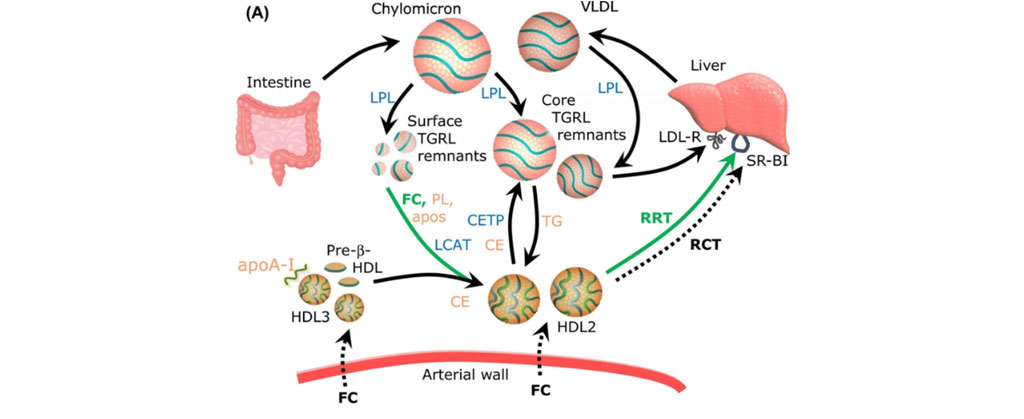
HDL prevents surface TRL remnants from accumulation in the arterial wall via acquiring phospholipid (PL) and free cholesterol (FC) during TG lipolysis (Kontush A, Trends in Molecular Medicine, 2020)
Importantly, our research has established that the capacity of HDL to acquire free cholesterol during lipolysis of TGRL by LPL is equally compromised in subjects displaying extremely high levels of HDL-C (PMID: 31840535). This functional deficiency probably reflects structural and compositional alterations of HDL in such subjects, involving extreme enlargement of HDL particles and their saturation with lipid molecules. This concept was published as a review entitled “HDL and Reverse Remnant-Cholesterol Transport (RRT): Relevance to Cardiovascular Disease” in Trends in Molecular Medicine (PMID: 32861590).
In short, the concept can be summarized as follows. CV diseases predominantly result from atherosclerosis, a natural biological phenomenon reflecting food intake and energy production in humans. Lipolysis of plasma TGRLs by LPL is an essential element of energy production that delivers free fatty acids to peripheral cells. HDL plays a key role in this process by acquiring surface lipids, including free cholesterol, that are released upon TGRL lipolysis. According to the reverse remnant-cholesterol transport (RRT) hypothesis, such removal of cholesterol from remnant lipoproteins followed by transport to the liver and excretion into the bile represents a major biological function of HDL that is essential for energy production, and which can reduce cholesterol influx into the arterial wall by accelerating the removal of atherogenic TGRL remnants from the circulation.
AXIS 3. To identify new risk factors in ASCVD.
Familial hypercholesterolemia. Familial hypercholesterolemia (FH) increases the incidence of ASCVD to 1%/year and the frequency of coronary artery diseases before 35 years old (PMID: 32964393). Some elements suggest that this may also be led by an exposition of the fetus to increased levels of cholesterol in utero (PMID: 28935756). Among them, increased number and size of fatty streaks have been reported in fetuses of pregnant women with hypercholesterolemia (PMID: 9389731), whose persistence may facilitate atherosclerosis development (PMID: 11133201). This association has also been observed in preclinical mouse models exposed during their fetal periods to high levels of cholesterol (PMID: 28935756; PMID: 12451021). However, underlying mechanisms are still not fully understood. These models are deeply explored in the lab to decipher early mechanisms and potential biomarkers of emerging disease.
In FH, the phenotype of genetically elevated LDL-C drives the clinical diagnosis but the trajectory toward ASCVD varies substantially. We have developed several approaches to finely characterize patients with severe hypercholesterolemia in order to better evaluate their cardiovascular risk. In a retrospective study of more than 4500 patients with an LDL-C above 1.9 g/L, we showed that only an LDL-C cut-off of 2.9 g/L was discriminant of monogenic FH. Below this level, hypercholesterolemia is at least in part, driven by a polygenic component that can be assessed by gene risk score (GS) analysis (PMID: 23433573). We combined the biochemical characteristics and the GS to developed a highly discriminant index. It allowed us to objectively distinguish monogenic from polygenic FH, unravel particular FH with complex “hidden” mutations. Moreover, this new ranking index is predictive of ASCVD risk evaluation in FH (PCT/FR2024/051639)
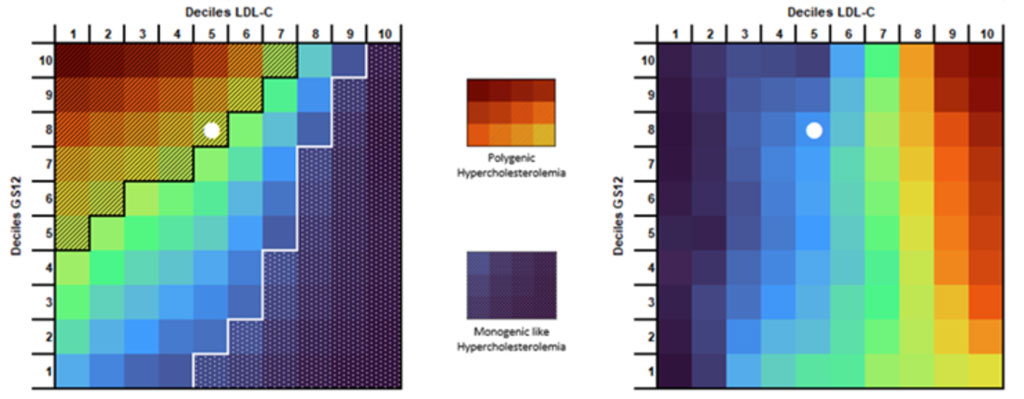
A new score for predicting ASCVD risk in Familial Hypercholesterolemia (PCT/FR2024/051639).
Myocardial infarction. For more than 50 years, a low HDL-C level has been recognized as an independent risk factor for cardiovascular disease. More recently, epidemiological studies have demonstrated that extremely high HDL-C concentrations are also associated with increased cardiovascular mortality, reflecting a U-shaped relationship between mortality and circulating HDL-C concentrations. In addition, failure of therapeutic trials designed to increase HDL-C have raised the concept of quality versus quantity, meaning that the physiological role of HDL particles is more important than their circulating concentration. Among the multiple biological activities carried by HDL, the capacity of HDL to promote macrophage cholesterol efflux was reported in several clinical studies to account for the protective action of HDL in CVD. Evaluation of this metric of HDL function in a large cohort of patients hospitalized for an acute myocardial infarction revealed that serum cholesterol efflux capacity was inversely associated with all-cause mortality in those patients independently of HDL cholesterol level and other risk factors (PMID: 30573028). This study supported the notion that HDL-based therapy could be beneficial in patients with myocardial infarction although the exact mechanism underlying this effect is not yet elucidated. Macrophage cholesterol efflux to HDL is inversely linked to inflammation through its repressive action on the NLRP3 inflammasome machinery and the subsequent secretion of interleukin-1β. Inhibition of this inflammatory pathway was associated with a reduced risk of recurrent cardiovascular events in stable patients with previous myocardial infarction. We reported that measurement of interleukin-1β at admission in patients with acute myocardial infarction was independently associated with the risk of mortality and recurrent major adverse cardiovascular events (PMID: 32861811). More recently we demonstrated that the combination of two biomarkers allows a better identification of patients at higher risk of recurrent cardiovascular events in the year following myocardial infarction. Patients combining HDL-mediated defective CEC and elevated inflammation measured by IL-1β levels are at very high residual cardiovascular risk after a recent MI suggesting a synergistic prognostic value of these biomarkers. The mechanisms of this higher risk could be explained by the presence of HDL particles exhibiting a specific defective ability to mediate cholesterol efflux and enhanced inflammatory properties. Indeed, high risk patients are characterized by dysfunctional HDL particles with reduced cholesterol efflux capacity and altered anti-inflammatory activities in macrophages and endothelial cells contributing to the elevation of the cardiovascular residual risk (see Figure). Our data underscore the clinical relevance of combining measurements of IL-1β and CEC to better identify STEMI patients at higher risk of recurrent MACE. A ‘dual pathway’ approach targeting inflammation and CEC with targeted therapies may have the potential to improve clinical outcome of coronary patients at high cardiovascular risk (PMID: 39506545).
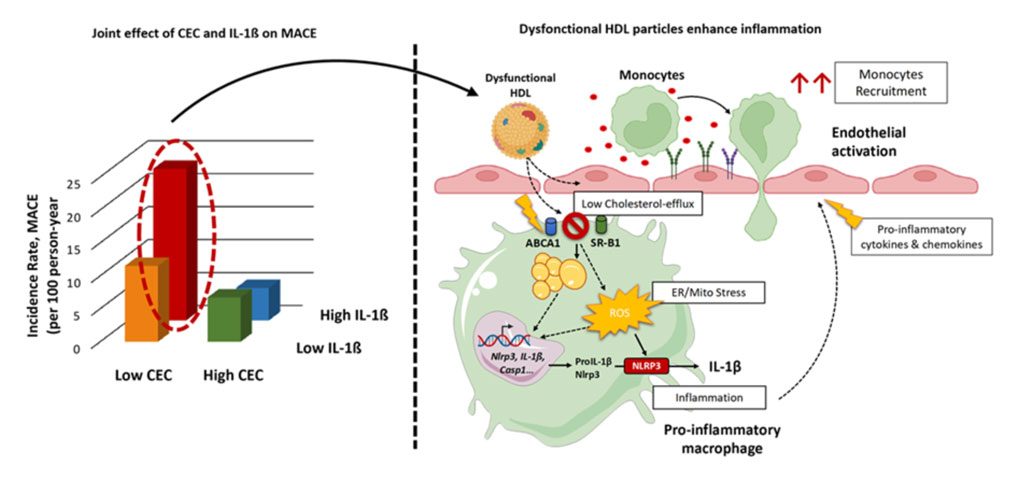
Impaired cholesterol efflux capacity and elevated IL-1β synergistically increase the residual cardiovascular risk in MI patients (Silvain J. & al., European Journal of Preventive Cardiology, 2024)
Immune diseases.
Alteration of lipid metabolism is frequently observed in numerous pathologies as secondary clinical manifestation of the disease. Such a disturbance is associated with autoimmune or inflammatory diseases, such as rheumatoid arthritis (RA), Systemic Lupus Erythematosus (SLE), Crohn’s disease, or sepsis, which are characterized by chronic inflammation and a high risk of developing cardiovascular disease (CVD) that is not fully explained by the classical Framingham risk factors. Our recent work uncovers the contribution of lipidomic and metabolic alterations to the presence of coronary artery calcium and the associated risk of coronary heart disease in SLE. Importantly, we identified novel lipid and metabolite biomarkers that may help stratify patients for reducing CVD morbidity and mortality in SLE (PMID: 37725854).
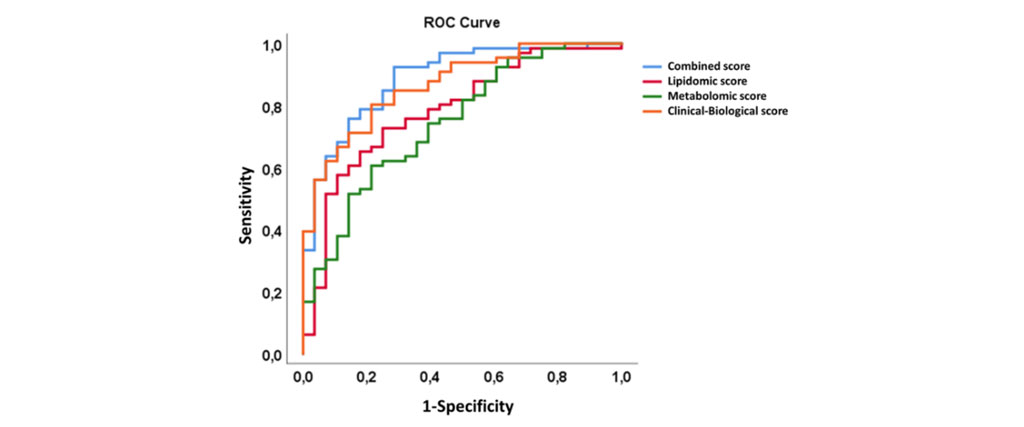
Prediction scores for the presence of coronary artery calcium in SLE (Urbain F. & al., EBioMedicine, 2023)
Immunity and cholesterol metabolism.
Our recent research suggests that mucosal-associated invariant T (MAIT) cells may play a role in protecting against ASCVD by promoting the excretion of cholesterol, particularly very-low-density lipoprotein (VLDL) cholesterol, through the intestines. This process is driven by IL-22 signaling, which increases cholesterol removal by upregulating ABCB1 in intestinal cells. We demonstrate in both animal models and a small group of humans with high cholesterol that higher MAIT cell levels are associated with healthier cholesterol profiles, characterized by lower VLDL and higher HDL. Conversely, people with higher BMI tend to have fewer MAIT cells, suggesting a link between MAIT cell depletion and poorer cardiometabolic health (PMID: 40135347).
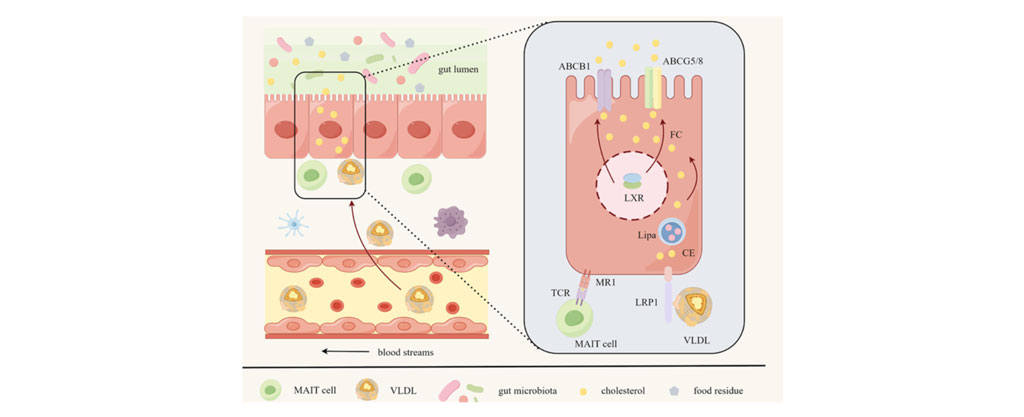
MAIT Cells Promote Cholesterol Excretion Pathways Mitigating Atherosclerosis. (Wang H. & al., Circulation Research, 2025)

| Name | Position | ORCID |
|---|
- Jalil, A, Pilot, T, Bourgeois, T, Laubriet, A, Li, X, Diedisheim, M et al.. Plasmalogen remodeling modulates macrophage response to cytotoxic oxysterols and atherosclerotic plaque vulnerability. Cell Rep Med. 2025;6 (5):102131. doi: 10.1016/j.xcrm.2025.102131. PubMed PMID:40345182 PubMed Central PMC12147908.
- Chemello, K, Guedon, AF, Techer, R, Jaafar, AK, Guilhen, S, Swietek, MJ et al.. Apolipoprotein E Plasma Concentrations Are Predictive of Recurrent Strokes: Insights From the SPARCL Trial. J Am Heart Assoc. 2025;14 (9):e036630. doi: 10.1161/JAHA.124.036630. PubMed PMID:40240927 PubMed Central PMC12184613.
- Wang, H, Kc, P, Zhang, K, Materne, C, Lhomme, M, Galier, S et al.. MAIT Cells Promote Cholesterol Excretion Pathways Mitigating Atherosclerosis. Circ Res. 2025;136 (9):968-981. doi: 10.1161/CIRCRESAHA.124.325841. PubMed PMID:40135347 .
- Le Goff, W, Bourron, O, Materne, C, Galier, S, Phan, F, Tan-Chen, S et al.. Inverse relationship between circulating sphingosine-1-phosphate and precursor species and coronary artery calcification score in type 2 diabetes. Cardiovasc Diabetol. 2025;24 (1):85. doi: 10.1186/s12933-025-02624-9. PubMed PMID:39984928 PubMed Central PMC11846453.
- Dahik, VD, Kc, P, Materne, C, Reydellet, C, Lhomme, M, Cruciani-Guglielmacci, C et al.. ABCG1 orchestrates adipose tissue macrophage plasticity and insulin resistance in obesity by rewiring saturated fatty acid pools. Sci Transl Med. 2024;16 (777):eadi6682. doi: 10.1126/scitranslmed.adi6682. PubMed PMID:39661702 .
- Silvain, J, Materne, C, Zeitouni, M, Procopi, N, Guedeney, P, Brugier, D et al.. Defective biological activities of high-density lipoprotein identify patients at highest risk of recurrent cardiovascular event. Eur J Prev Cardiol. 2025;32 (13):1273-1285. doi: 10.1093/eurjpc/zwae356. PubMed PMID:39506545 .
- European Atherosclerosis Society Familial Hypercholesterolaemia Studies Collaboration (EAS FHSC). Association of BMI, lipid-lowering medication, and age with prevalence of type 2 diabetes in adults with heterozygous familial hypercholesterolaemia: a worldwide cross-sectional study. Lancet Diabetes Endocrinol. 2024;12 (11):811-823. doi: 10.1016/S2213-8587(24)00221-3. PubMed PMID:39374602 .
- Watts, GF, Rosenson, RS, Hegele, RA, Goldberg, IJ, Gallo, A, Mertens, A et al.. Plozasiran for Managing Persistent Chylomicronemia and Pancreatitis Risk. N Engl J Med. 2025;392 (2):127-137. doi: 10.1056/NEJMoa2409368. PubMed PMID:39225259 .
- Galier, S, Darabi, M, Ma, F, Materne, C, Guillas, I, Le Goff, W et al.. Reduced Capacity of High-Density Lipoprotein to Acquire Free Cholesterol From Triglyceride-Rich Lipoproteins Is Associated With Elevated Postprandial Hypertriglyceridemia in Healthy Men. J Am Heart Assoc. 2024;13 (15):e034770. doi: 10.1161/JAHA.123.034770. PubMed PMID:39082393 PubMed Central PMC11964020.
- Palmulli, R, Couty, M, Piontek, MC, Ponnaiah, M, Dingli, F, Verweij, FJ et al.. CD63 sorts cholesterol into endosomes for storage and distribution via exosomes. Nat Cell Biol. 2024;26 (7):1093-1109. doi: 10.1038/s41556-024-01432-9. PubMed PMID:38886558 .
- Béliard, S, Saheb, S, Litzler-Renault, S, Vimont, A, Valero, R, Bruckert, É et al.. Evinacumab and Cardiovascular Outcome in Patients With Homozygous Familial Hypercholesterolemia. Arterioscler Thromb Vasc Biol. 2024;44 (6):1447-1454. doi: 10.1161/ATVBAHA.123.320609. PubMed PMID:38695169 .
- European Atherosclerosis Society Familial Hypercholesterolaemia Studies Collaboration. Familial hypercholesterolaemia in children and adolescents from 48 countries: a cross-sectional study. Lancet. 2024;403 (10421):55-66. doi: 10.1016/S0140-6736(23)01842-1. PubMed PMID:38101429 .
- Guillas, I, Lhomme, M, Pionneau, C, Matheron, L, Ponnaiah, M, Galier, S et al.. Identification of the specific molecular and functional signatures of pre-beta-HDL: relevance to cardiovascular disease. Basic Res Cardiol. 2023;118 (1):33. doi: 10.1007/s00395-023-01004-2. PubMed PMID:37639039 .
- Bosteels, V, Maréchal, S, De Nolf, C, Rennen, S, Maelfait, J, Tavernier, SJ et al.. LXR signaling controls homeostatic dendritic cell maturation. Sci Immunol. 2023;8 (83):eadd3955. doi: 10.1126/sciimmunol.add3955. PubMed PMID:37172103 .
- Paterson, HAB, Yu, S, Artigas, N, Prado, MA, Haberman, N, Wang, YF et al.. Liver RBFOX2 regulates cholesterol homeostasis via Scarb1 alternative splicing in mice. Nat Metab. 2022;4 (12):1812-1829. doi: 10.1038/s42255-022-00681-y. PubMed PMID:36536133 PubMed Central PMC9771820.
- Darabi, M, Lhomme, M, Dahik, VD, Guillas, I, Frisdal, E, Tubeuf, E et al.. Phosphatidylserine enhances anti-inflammatory effects of reconstituted HDL in macrophages via distinct intracellular pathways. FASEB J. 2022;36 (5):e22274. doi: 10.1096/fj.201800810R. PubMed PMID:35416331 .
- Gallo, A, Giral, P, Rosenbaum, D, Mattina, A, Kilinc, A, Giron, A et al.. Myocardial fibrosis assessed by magnetic resonance imaging in asymptomatic heterozygous familial hypercholesterolemia: the cholcoeur study. EBioMedicine. 2021;74 :103735. doi: 10.1016/j.ebiom.2021.103735. PubMed PMID:34864619 PubMed Central PMC8646177.
- Aubart, FC, Poupel, L, Saint-Charles, F, Charlotte, F, Arsafi, Y, Frisdal, E et al.. Profound systemic alteration of the immune phenotype and an immunoglobulin switch in Erdheim-Chester disease in 78 patients from a single center. Haematologica. 2022;107 (6):1347-1357. doi: 10.3324/haematol.2021.279118. PubMed PMID:34647443 PubMed Central PMC9152974.
- EAS Familial Hypercholesterolaemia Studies Collaboration (FHSC). Global perspective of familial hypercholesterolaemia: a cross-sectional study from the EAS Familial Hypercholesterolaemia Studies Collaboration (FHSC). Lancet. 2021;398 (10312):1713-1725. doi: 10.1016/S0140-6736(21)01122-3. PubMed PMID:34506743 .
- Lécuyer, E, Le Roy, T, Gestin, A, Lacombe, A, Philippe, C, Ponnaiah, M et al.. Tolerogenic Dendritic Cells Shape a Transmissible Gut Microbiota That Protects From Metabolic Diseases. Diabetes. 2021;70 (9):2067-2080. doi: 10.2337/db20-1177. PubMed PMID:34078628 PubMed Central PMC8576430.
21. Impaired metabolism predicts coronary artery calcification in women with systemic lupus erythematosus. Urbain F, Ponnaiah M, Ichou F, Lhomme M, Materne C, Galier S, Haroche J, Frisdal E, Mathian A, Durand H, Pha M, Hie M, Kontush A, Cluzel P, Lesnik P, Amoura Z, Guerin M, Cohen Aubart F, Le Goff W. EBioMedicine. 2023 Oct;96:104802. doi: 10.1016/j.ebiom.2023.104802. Epub 202
Cholesterol, Lipoproteins, and Cardiovascular Health: Separating the Good (HDL), the Bad (LDL), and the Remnant
Author: Anatol Kontush
Publisher: Wiley & Sons
Publication date: October 22, 2024
Language: English
Print length: 384 pages
ISBN-10: 1394158378
ISBN-13: 978-1394158379
High-Density Lipoproteins – Structure, Metabolism, Function, and Therapeuthics
Authors: Anatol Kontush, M. John Chapman
Publisher: Wiley & Sons
Publication date: February 20, 2012
Language: English
Print length: 648 pages
ISBN-10: 0470408219
ISBN-13: 978-0470408216
1. Classification of severe hypercholesterolemias (PCT/FR2024/051639).
2. Drug repositioning for use in the treatment and/or prevention of atherosclerosis (EP24305179).
3. Transcription factor ZNF471 as a therapeutic agent and a biomarker (PCT/EP2018/073545).
4. Novel assay of HDL function (PCT/EP2018/071172).
5. Reconstituted high density lipoproteins composition and uses thereof (PCT/EP2014/070970).
6. The ABCG1 gene as a marker and a target gene for treating obesity (PCT/EP2011/073140).




